
But all of these valence-bond models, as they are generally called, are very limited in their applicability and predictive power, because they fail to recognize that distribution of the pooled valence electrons is governed by the totality of positive centers. 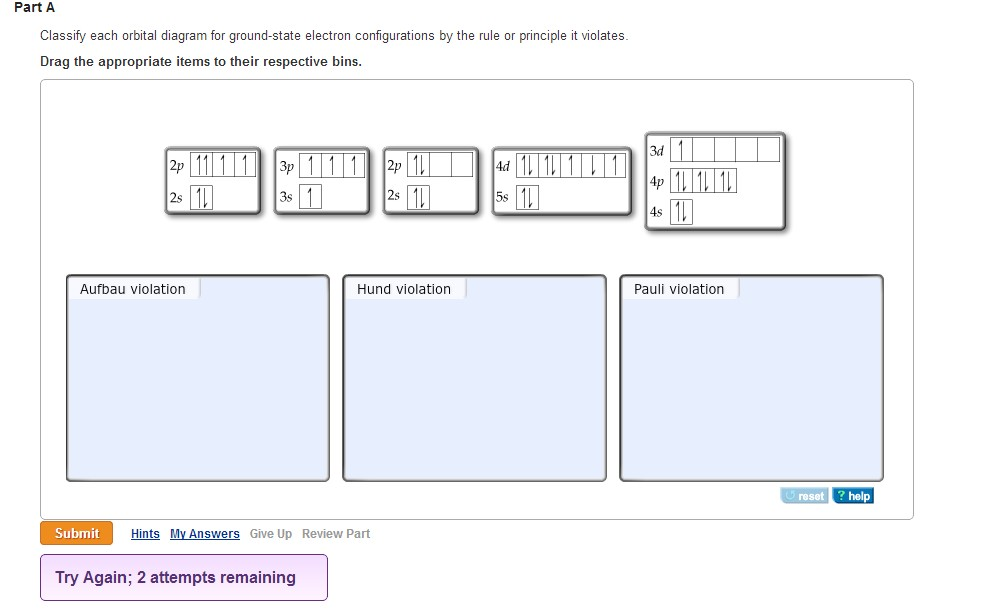
The more sophisticated hybridization model recognized that these orbitals will be modified by their interaction with other atoms. This is a big departure from the simple Lewis and VSEPR models that were based on the one-center orbitals of individual atoms. Welcome to the world of molecular orbitals ! The sculpting itself is created by the positive cores that remain after the "atoms" in this molecule have been stripped of their valence electrons, which are now able to range over the entire molecule, occupying orbitals whose spatial extent and properties depend entirely on the charges and geometric distribution of the collection of positive centers. The colors of the sculpted folds represent different electron densities- probabilities of finding an electron at any given point. Look at the computer-generated model of the nicotine molecule shown at the upper right corner of this window. Note: this document will print in an appropriately modified format (? pages) Nuclear charge: As the nuclear charge increases, electronegativity increases.⇐index | properties | models | covalent | polar | geometry | hybrid 1 | hybrid 2 molecular orbitals | coordination complexes | metals-semiconductors This is because the electrons are far from the nucleus, and there is a lesser force of attraction. Atomic size: As the atomic size increases, electronegativity decreases. The tendency of an atom in a molecule to attract the shared pair of electrons towards itself is called its electronegativity.Įlectronegativity values also depend on the following factors: As atomic size increases, the nucleus’s attraction to the incoming electron lessens, and the electron gain enthalpy becomes less negative. However, increasing atomic size is far more pronounced than the effect of increasing nuclear charge. Variation within a group: The atomic size and nuclear charge increase as we move down a group. Both of these factors increase the nucleus’s attraction to the incoming electron, and as a result, the electron gain enthalpy decreases from left to right.ī. Variation along a period: The atomic size decreases, and the nuclear charge increases as we walk across a period from left to right. Variation of Electron Gain enthalpy in the periodic tableĪ. Because the s-subshell can only have two electrons, this block comprises only two groups \(\left(\) The elements in the s-block of the periodic table have the last electron filled in valence s-sub-shell of the outermost energy shell. 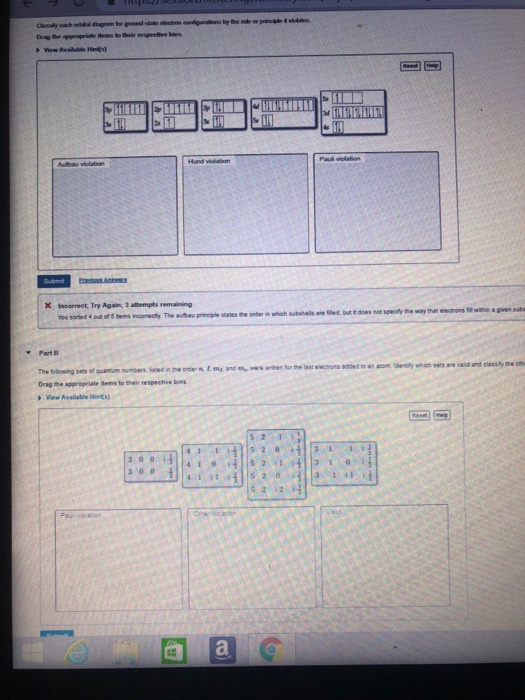
The long form of the periodic table is divided into four blocks known as the s, p, d and f-blocks. Periodic Classification of Elements Based on Electronic Configuration The properties of elements and their compounds can be predicted based on their position in the periodic table.It is simpler to study, compare, and distinguish the properties of elements and compounds from different groups.Classification assists us in understanding the properties of elements and their compounds.Some of the advantages of classification of elements are given below: Elements are the basic units of all matter.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
